| Cancer Care Ontario's Data Book - 2016-2017 |
|
Updated in February 2015
(Please Note: This section is under review and will be updated soon)
Has NACRS data been considered for use in the new funding model?
What are CCO’s expectations around the ALR Transition implementation timelines?
What data is being used to support funding in the current fiscal year (2013/14)?
For OPIS sites, can consult data just be built into the OPIS application?
What are the reporting requirements for the 'MIS Functional Centre' data element?
What are the reporting requirements for the 'Clinical Trials Flag'?
What are the reporting requirements for the 'Inpatient Flag'?
What formula should be used to calculate/report Body Surface Area (BSA)?
For any type of follow-up, how is that activity going to be distinguished from the initial consult?
Will the clinic file need to include the PSO information that is scheduled for full RCC’s this year?
Are BCG regimens for urology patients required to be reported to ALR?
Can there be more than one value reported for intent of treatment (e.g. curative and adjuvant)?
Do blood products (e.g. IVIG, platelets, pRBC) need to be reported?
1. Has NACRS data been considered for use in the new funding model?
Yes, NACRS was considered as a potential data source for supporting the new Systemic Treatment Funding Model. Upon review it was determined that a richer data set (than available from NACRS) was required to support the new patient-based funding model. Additionally, it was determined that the data source for the funding model should be a system owned and maintained by Cancer Care Ontario, allowing for more flexibility to develop and refine the data collected as the funding model advances.
The MDS currently only captures clinic data for new cases that had their first consult date on or after April 1st, 2011. No systemic treatment or follow up assessment data is reported through MDS. ALR Data includes data elements that constitute patient level activity that are collected at hospitals who deliver systemic treatments as well as outpatient oncology clinics (i.e. initial consults and follow up assessments). Please refer to the ALR Data Package for full information on the additional data elements required for facilities submitting MDS.
While facilities that have implemented OPIS are submitting all required treatment data as part of their STCPOE agreement, the transition to ALR will require data collection around outpatient oncology clinics (i.e. initial consults and follow up assessments). As OPIS is designed to capture treatment data, consults and follow-up care are not captured in OPIS. Note: Consult and follow-up data is also required for patients who do not proceed to have systemic treatment following their initial consultation with a medical oncologist. Please refer to the ALR Data Package for full information on the additional data elements required for facilities submitting MDS & ALR (sys)/OPIS.
The Minimum Data Set (MDS) is a sub-set of the ALR data set, used for calculating Systemic Treatment Wait Times. Once a facility has fully transitioned to full ALR reporting, the wait times can be calculated from the ALR submission and the facility will no longer need to submit a separate MDS file.
CCO is working to determine the best mechanism for reimbursing hospitals for treatment bundles. Depending on the solution selected, there may be some overlap between eClaims and ALR. More information will be provided to sites once this issue has been resolved.
6. What are CCO’s expectations around the ALR Transition implementation timelines?
a. By when are facilities required to submit Test data?
To allow for sufficient testing of data and metrics, it would be ideal if initial test submissions could be completed in early fall of FY 2013/2014.
b. By when are facilities required to submit Production data?
To support the new systemic funding model, April 2014 data would need to be collected and submitted to production by June 2014.
7. What data is being used to support funding in the current fiscal year (2013/14)?
For fiscal year 2013/14, volumes will be counted as they were counted in 2012/13, i.e. there are no new data collection requirements.
No, while the facility would be submitting all required treatment data, we would still require additional data elements around initial consults and follow up assessments to be submitted (a full clinic visit file, as outlined below). Please refer to the ALR Data Package for full information on the additional data elements required.
Additional data elements required for facilities currently submitting MDS & ALR(sys)/OPIS
Visit Time
HCP Number
Visit Program Code Inpatient Flag
Disease Sequence Number
MIS Functional Centre Patient Postal Code
Visit Type
Radiation Review Flag
Ideally the ALR Transition project would align to your facility’s STCPOE implementation timelines. The Systemic Treatment Information Program (STIP) team at CCO has expertise in STCPOE implementation and maintains a Community of Practice around STCPOE. Please contact CCO if you would like to explore what help we may be able to offer.
10. What is the process for ALR data package submissions?
a) The required data for the ALR submission is extracted from the local data system(s) and formatted into CSV files. These files are packaged into a ZIP archive file, which is the ALR Data Package.
b) The ALR Data Package is uploaded to CCO via the Data Book SSL web upload application.
c) The ZIP file is validated, and the contained CSV files are extracted, validated, and loaded into the CCO database.
d) QA reports from the load are generated and posted to the CCO SSL web site for download.
11. For OPIS sites, can consult data just be built into the OPIS application?
OPIS is used to prescribe and administer systemic treatment. OPIS does not support clinic activities prior to treatment and there are no plans to develop such functionality. As such, there is no data on pre-treatment clinic activities within OPIS. Data elements generated during treatment and captured by ALR can be captured through OPIS. The pre-treatment data elements must be generated either from the hospital’s EMR system or manually from the patient chart.
A baseline file is used to identify all existing patient cases prior to the hospital’s start of data submission (for Clinic Visits). Once the visit activity submission commences, each visit is cross referenced back to the baseline file to determine if the patient is a new case (as of the visit date). If the patient does not match a record on the baseline file, it is counted as a new case visit and subsequently added onto the file to identify further follow up visits. A record match will count as a follow up visit. New case and follow up visits are grouped by the Visit Program Code.
13. What is required for the baseline file?
Process for Preparing the Baseline:
I. CCO will generate a baseline file based on current MDS data, going back to April 2011.
II. If there are issues with the baseline file, any missing or required updates should be submitted as per a regular MDS resubmission.
III. Steps 1 & 2 will be repeated until the facility is confident with the quality of the baseline file.
IV. Based on the date range that has been decided upon for the site’s ALR Test Metrics submission, CCO will generate a baseline file that ends one month before the date range for Test Metrics.
V. Prior to facilities submitting to production in 2014, a baseline file for Production ALR will be generated representing data from April 2011 – March 2014.
14. Are facilities required to report Disease Sequence Number?
The disease sequence number element is optional. However, Disease Sequence Number must either be entered or left blank on ALL records in ALL entities from a submitting site, and that must remain consistent from month to month.
If left blank, CCO will derive a value from the Registration Date. If this is the case, the field must be unique amongst Diseases within the Patient.
15. What are the reporting requirements for 'Visit Time'?
This should be appointment time, not discharge time. This field is used to identify multiple visits on the same day.
16. What are the reporting requirements for the 'MIS Functional Centre' data element?
The MIS Functional Centre is an optional field and therefore NULL is a valid submission.
If hospitals choose to report the MIS functional center they should report the functional center as per their MIS submission to MOHLTC. Therefore if the hospital submits data to both the 7134066 functional center for treatment activity and the 7130566 for clinic activity when submitting MIS data, they should report this accordingly to CCO Data Book.
After consulting with the Ontario Cancer Registry (OCR) team at CCO, hospitals are required to report the following: patient's permanent postal code at time of registration.
The current ALR definition for Patient Postal Code at Diagnosis will be revised to say postal code at time of registration.
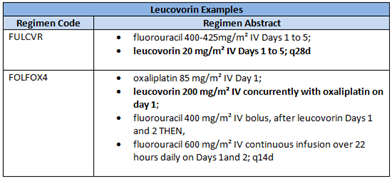
18. What are the reporting requirements for CCO Regimen?
CCO regimens are available in appendix 1.22 of ALR Databook.
Facilities can request to have regimens added to the list and updates are made on a quarterly basis. As funding for systemic treatment will be based on evidence-informed practice (starting 2014/15), hospital regimen codes need to be mapped to the CCO regimen codes.
The Oral Chemo Clinic Visit (‘OC’) flag for the ‘Visit Type’ element should be used to track clinic visits (i.e. follow-up visits) that are specific to oral chemo only. If PARENTERAL treatment is also part of the associated regimen, the parenteral treatment takes precedence over the oral treatment and the field should be left blank.
With regard to reporting, the hierarchy should be as follows:
- BLANK/NULL (parenteral in person visit)
- OC (Oral chemotherapy visit)
- OM (Parenteral patient Interaction by Phone)
20. What are the reporting requirements for 'Line of Therapy'?
The line of therapy is specific to chemotherapy and does not include other treatments/procedures such as surgery.
Any unique physician identifier that is numeric will work for this field. The HCP number does not have to be unique for your facility as it is used in conjunction with the Submitting Hospital Number as a key identifier for that table.
The purpose of this element is to uniquely identify HCPs and the value reported is determined by the facility. If Nurse Practitioners are completing follow-up visits the Nurse Practitioners HCP number should be included.
If both Nurse Practitioner and Oncologist are involved in the clinic visit, the oncologist should be reported.
For level 3 ALR hospitals, the current scope of reporting only includes the Systemic (‘Sys’) program code (unless the facility also delivers radiation) and as such this field may be hard-coded.
However, facilities should be aware that in the future, there may also be a need to report other programs such as 'palliative care' and 'supportive care'.
As Cancer Care Ontario develops the Systemic Treatment Funding Model in direction from the Ministry of Health and Long Term Care, additional programs or data elements may be required to support the funding model.
24. What are the reporting requirements for the 'Clinical Trials Flag'?
In order to ensure the clinical trials are appropriately funded and supported by Cancer Care Ontario, a Clinical Trials Working Group has been launched as part of the development of the new Systemic Treatment Funding Model.
All clinical trials are to be reported to ALR and identified as clinical trials using the 'Clinical Trials Flag'.
The Clinical Trials Working Group is working to draft a clear definition as to what constitutes a clinical trial so they are consistently reported across the province. A briefing note outlining this definition will be distributed by Cancer Care Ontario as soon as possible.
25. What are the reporting requirements for the 'Inpatient Flag'?
Systemic Treatment inpatients are out of scope for the Systemic Treatment Funding Model, for now. Facilities have two options for reporting inpatients:
I. Report inpatients to ALR and identify them using the ‘inpatient flag’
II. Do not report inpatients to ALR
Please note that if/when inpatients are in scope for the Systemic Treatment Funding Model, the reporting requirements for inpatients may change.
26. What formula should be used to calculate/report Body Surface Area (BSA)?
CCO released a position statement on BSA calculation that promotes the use of the Mosteller equation. Please disregard the comment in the-online Data Book that mentioned MEDLOG. This will be updated for the next release.
All of the regimens listed in the KTE package should be in Appendix 1.22 of Data Book. However, there may be parental treatments in the CCO regimen list that are not in the KTE package. The KTE package only includes a list of evidence informed regimens and disease groups for the Parenteral Treatment- Adjuvant, Curative, and Neo-Adjuvant bundle. Evidence informed regimens for the palliative and oral treatment bundles are still being collected.
For more details on Regimens please visit CCO drug formulary website: http://www.cancercare.on.ca/toolbox/drugs/drugformulary/
Clinic visit activity (seeing the medical oncologist in the clinic), would be captured under the ALR clinic visit file. When the patient returns the following day to the chemotherapy suite for treatment, this activity would be captured in the ALR Systemic Drug Delivery Event File. In the case where the patient sees the medical oncologist in clinic and then receives treatment in the chemotherapy suite on the same day, this activity would be captured in and submitted in the two separate files.
29. Does a clinical trial count as a line of therapy?
Yes, a clinical trial counts as a line of therapy.
Yes, a change in treatment due to intolerance/side effects would be considered a line of therapy. A change in treatment due to disease progression would also be considered a line of therapy.
Yes, if a patient does not receive a complete course of therapy due to loss of follow-up or for unknown reasons, this would be considered a line of therapy. Please note, a new line of therapy is when a patient starts a new treatment/regimen, and as such transitioning from part a to part b of a regimen protocol is not a new line of therapy.
32. Are out of office consults to be reported to ALR?
Out of office consults should be reported to ALR so that hospitals may be funded accordingly. It is appropriate for hospitals to report the consult date at the time of the initial registration. A briefing note will be released to further address this requirement.
That is correct; this field is intended to identify the method of contact for clinic visits. If the calls are not replacing clinic visits, they would not need to be reported.
Note: this flag is only applicable if it’s a parenteral visit and takes place on the call. Most important is to identify whether parenteral (leave null) or oral chemo.
Both initial consults and follow-ups should be submitted to CCO in the same fashion. There is no specific data element that is used to distinguish the two. Our methodology validates each clinic visit file that is submitted by comparing it against previous clinic visits for the same case. The activity is then recorded as a new consult or follow-up based on whether there is a previous record.
The PSO component of the clinic visit file is not mandatory for non-RCC hospitals in 2014/15. However, hospitals can submit this if they choose to do so. Please note that PSO best practice is being incorporated into the new funding model for systemic treatment and PSO reporting requirements may change in future years. We understand that this is not necessarily a definitive answer, but Quality Based Procedures are largely driven by policy set by the Ministry of Health and, for the most part, policies need to be consistent across all Quality Based Procedure funding models.
In this case, the referral date should be the same as the first consultation date. The expectation for all hospitals is that inpatients should be seen within 24 hours.
To further support this question, here are two scenarios:
a) A patient is admitted to the hospital; however, the oncologist notes that they are not ready for treatment. The patient is told to return in a month.
Assuming the patient was seen on the same day they were admitted, the referral to consult would be 0 days.
b) A patient is admitted to the hospital and begins systemic treatment on the inpatient floor, and is then discharged and transferred to the outpatient clinic.
Once again, assuming that the patient was seen by the medical oncologist on the same day that they were admitted, the referral to consult would be 0 days.
Intent of systemic treatment is only captured in the systemic drug delivery event file, when treatment is delivered. Consults and follow-up visits are captured in the clinic visit file and intent of systemic treatment is not a data element in that file (i.e. intent of systemic treatment is not reported for consult or follow-up visits).
Intent of systemic treatment is to be determined by the medical oncologist. The definitions of the acceptable values (based on the NHS definitions) are:
· C = Curative: Where main aim is long-term survival.
· P = Palliative: Where main aim is to ameliorate symptoms.
· A = Adjuvant: An adjunct to potentially curative local treatment.
· N = Neoadjuvant: An adjunct given prior to potentially curative local treatment.
This example can be broken down into two scenarios:
Scenario A (new treatment intent + new regimen): In most cases the regimen would also change and so a new treatment intent and regimen would be reported at the time of reporting the new regimen.
Scenario B (new treatment intent + same regimen): In an extremely rare case the regimen may not change, the treatment intent should be updated to palliative.
How telephone follow-ups will be incorporated into the systemic treatment funding model is to be determined. Should funding be provided for telephone follow-ups then you would want to ensure that these are reported.
Ideally all clinic visits, including telephone follow-ups are reported to ALR in the clinic visit file.
The ‘Visit Type’ field in the clinic visit file is intended to identify the method of contact for clinic visits. The hierarchy for reporting this field is as follows:
- BLANK/NULL (parenteral in person visit)
- OC (Oral chemotherapy visit)
- OM (Parenteral patient Interaction by Phone)
We are currently exploring opportunities for clearer reporting requirements for ‘Visit Type’
41. Are BCG regimens for urology patients required to be reported to ALR?
BCG regimens are out of scope for the systemic treatment funding model even if/when completed in the systemic treatment suite. Urology patients receiving BCG regimens do not need to be reported to ALR.
42. What are the ALR reporting requirements for drugs?
All anti-cancer drugs must be reported along with the DIN and dose administered.
Bisphosphonates are considered active treatment and are to be reported to ALR for purposes of the systemic treatment funding model. Bisphosphonates such as Pamidronate, Zoledronic Acid, Denosumab & Clodronate, must be reported along with the DIN and dose administered.
Certain drugs, although not anti-cancer, are integral components of the regimens and these are to be reported along with the DIN and dose administered
· E.g. Mesna, although not anti-cancer, must be given with any chemotherapy involving cyclophosphamide and ifosfamide. Mesna needs to be reported along with the DIN and dose administered.
· E.g. Leucovorin, although not anti-cancer and not required to be given in combination with other drugs, can be an integral part of a regimen. Leucovorin needs to be reported along with the DIN and dose administered.

The following supportive/non-chemotherapeutic do not need to be reported:
• Antiemetic drugs, e.g. Aprepitant
• Symptom management drugs, e.g. Ranitidine
• Growth stimulating drugs, e.g. Filgrastim, Erythropoietin, Darbepoetin, Pegfilgrastim
The reporting requirements around hormonal treatments are still under review and information will be shared as soon as possible.
Yes, in this case using ‘Arrived but not Treated’ is appropriate.
Bisphosphonates are considered active treatment and are to be reported to ALR for purposes of the systemic treatment funding model. In this scenario, docetaxel & pamidronate would be reported separately, as two drugs:
1. Docetaxel
2. Pamidronate
45. Can there be more than one value reported for intent of treatment (e.g. curative and adjuvant)?
Intent of systemic treatment is to be determined by the medical oncologist. The definitions of the acceptable values (based on the NHS definitions) are:
• C = Curative: Where main aim is long-term survival.
• P = Palliative: Where main aim is to ameliorate symptoms.
• A = Adjuvant: An adjunct to potentially curative local treatment.
• N = Neoadjuvant: An adjunct given prior to potentially curative local treatment.
As the definition for adjuvant and neo-adjuvant is before or after potentially curative treatment, adjuvant would be reported as the line of therapy in the scenario where intent of treatment is both curative and adjuvant.
A chemotherapy regimen can be made up of multiple drugs. If this is the case, one record is required for each separate drug (with the same regimen code on each file).
A patient can also be receiving multiple drugs that are not part of one regimen. For example, a patient may be on the anti-neoplastic regimen Docetaxel and separately/concurrently on Pamidronate for bone metastases. In this scenario, as Bisphosphonates are considered active treatment and are to be reported to ALR for purposes of the systemic treatment funding model, Docetaxel & Pamidronate would be reported separately as two drugs. Again, one record is required for each separate drug, with different regimen codes (DOCE for docetaxel and PMDR for Pamidronate).
The systemic treatment funding model does not apply to systemic treatment for non-cancer diagnoses (e.g. chemotherapy for rehumatoid athristis). Funding would not be provided for these patients as there would not be a cancer diagnosis. Therefore, activity does not have to be reported.
Line of therapy applies to patients being treated with palliative intent. Both ‘null/blank’ and ‘0’ are accepted values in this field for patients being treated with adjuvant/neo-adjuvant/curative intent. Line of therapy is to be determined by the medical oncologist or hematologist. Line of therapy is specific to systemic treatment (including parenteral and oral), and does not include other treatments or procedures such as supportive drugs (e.g. bisphosphonates), hormonal*** treatments or surgery.
***Please note the following: In prostate cancer, a couple of new hormone therapies are now counted as a line in therapy (e.g., oral drugs abiraterone and enzalutamide)
In breast cancer, there has been some debate (though no further action) on whether or not hormonal agents (e.g. oral drug tamoxifen) should be classified as a line of therapy. Currently they are not considered a line of therapy.
Report the appropriate line of therapy as per the above guidance. It does not matter if the first line of therapy is not reported to ALR if it was delivered in an inpatient setting.
49. Does CCO offer ICD code training for facilities?
CCO currently does not offer ICD diagnosis training, although CCO is exploring the possibility of hosting some webinars on this in the future.
CCO has looked into this and the best resource appears to be the web based training available, free, from NCI/SEER in the States. Going section by section (through the different cancer sites), one of the modules in each section is ICD-O-3 coding. Here is the link to the main SEER page: http://seer.cancer.gov/ with useful information available in the tab called Information for Cancer Registrars.
Here is the link to the web training within the SEER site: http://training.seer.cancer.gov/
It is also important that anyone coding ICD-O-3 needs the ICD-O-3 coding manual as software and other types of topography and morphology pick lists do not contain all of the synonyms that are available within the manual.
Bisphosphonates are considered active treatment and are to be reported to ALR for purposes of the systemic treatment funding model. In this scenario docetaxel & pamidronate would be reported separately, as two drugs:
1. Docetaxel
2. Pamidronate
51. Do blood products (e.g. IVIG, platelets, pRBC) need to be reported?
No, blood products would not be reported in the ALR Systemic Drug Delivery Event file. Procedures related to blood, e.g. transfusions, would be reported in the AL Procedures file. The exact reporting requirements for the procedures file are under review. Specifically, reporting requirements for transfusions are being reviewed by the Systemic Treatment Hematology Working Group.
| View the Data Book FAQ |